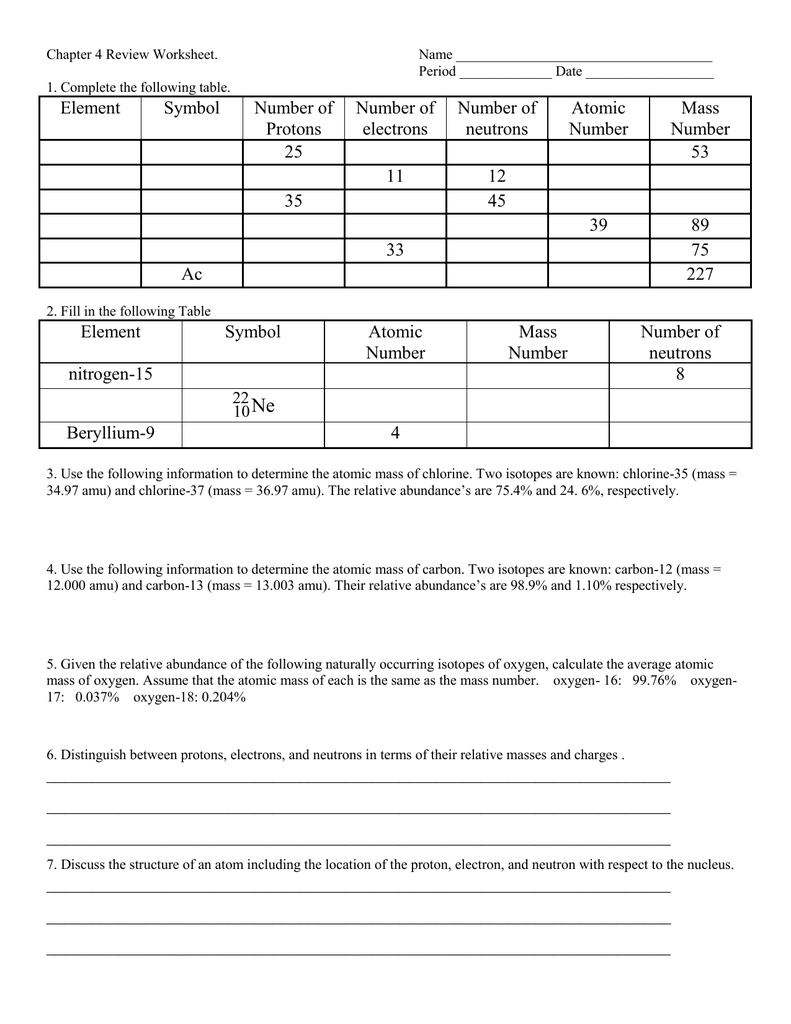
It can help you make sure you understand the fundamentals of the subject and can make sure you are prepared for any tests or exams. ConclusionĪn atomic structure review worksheet answer key can be a great tool for students who need to review the basics of atomic structure. Once you have found the right answer key, you can use it to review your knowledge of atomic structure and ensure you are prepared for any tests or exams. Additionally, you should make sure the answer key is designed to help you understand the material quickly so you can spend more time on solving problems. You should also look for a worksheet answer key that is easy to understand and provides helpful explanations of the material. 
You will want to make sure the answer key is up-to-date and accurate. Finding the Right Atomic Structure Review Worksheet Answer Keyįinding the right atomic structure review worksheet answer key can be a challenge. This can be useful when you are looking to solve complex problems. Additionally, an atomic structure review worksheet answer key can help you gain a better understanding of how the different parts of the worksheet interact with each other. This can be especially helpful if you are studying for a test or just need to refresh your knowledge of the subject. It can help you review the basics of atomic structure to make sure you understand it. Using an answer key for an atomic structure review worksheet can provide a number of benefits. Benefits of Using an Atomic Structure Answer Key With the right answer key, you can make sure you know what each part of the worksheet means and how to use it. An atomic structure review worksheet answer key can help you review the basic principles of atomic structure and make sure you understand them. It’s the basis of all matter, and understanding it is key to understanding the universe. Atomic Structure Review Worksheet Answer Keyįebru1 min read An Overview of Atomic Structure and its BasicsĪtomic structure is one of the most important topics in chemistry, physics, and biology. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Robinson, PhD, Klaus Theopold, Richard Langley We recommend using aĪuthors: Paul Flowers, Edward J. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, 
Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the (d) The paths followed by the α particles match the predictions from (a), (b), and (c). If the nucleus is larger, the positive charge is larger and the expected deflections are larger-more α particles will be deflected, and the deflection angles will be larger. (c) If the nucleus is smaller, the positive charge is smaller and the expected deflections are smaller-both in terms of how closely the α particles pass by the nucleus undeflected and the angle of deflection. (b) Higher-energy α particles that pass near the nucleus will still undergo deflection, but the faster they travel, the less the expected angle of deflection. The more directly toward the nucleus the α particles are headed, the larger the deflection angle will be. Those α particles that pass near the nucleus will be deflected from their paths due to positive-positive repulsion. (a) The Rutherford atom has a small, positively charged nucleus, so most α particles will pass through empty space far from the nucleus and be undeflected.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
